Background
On 16 December 2025, the European Commission published its long-awaited Proposal for a Biotech Act with an aim to “build a world-leading health biotech industry” and transform Europe into a “Biotech Powerhouse”. The Proposal recognises that the EU is losing ground to global competitors such as the US and China in developing innovative biological products and identifies insufficient funding, regulatory bottlenecks, and barriers to innovation as key obstacles limiting the EU’s ability to compete in this space.
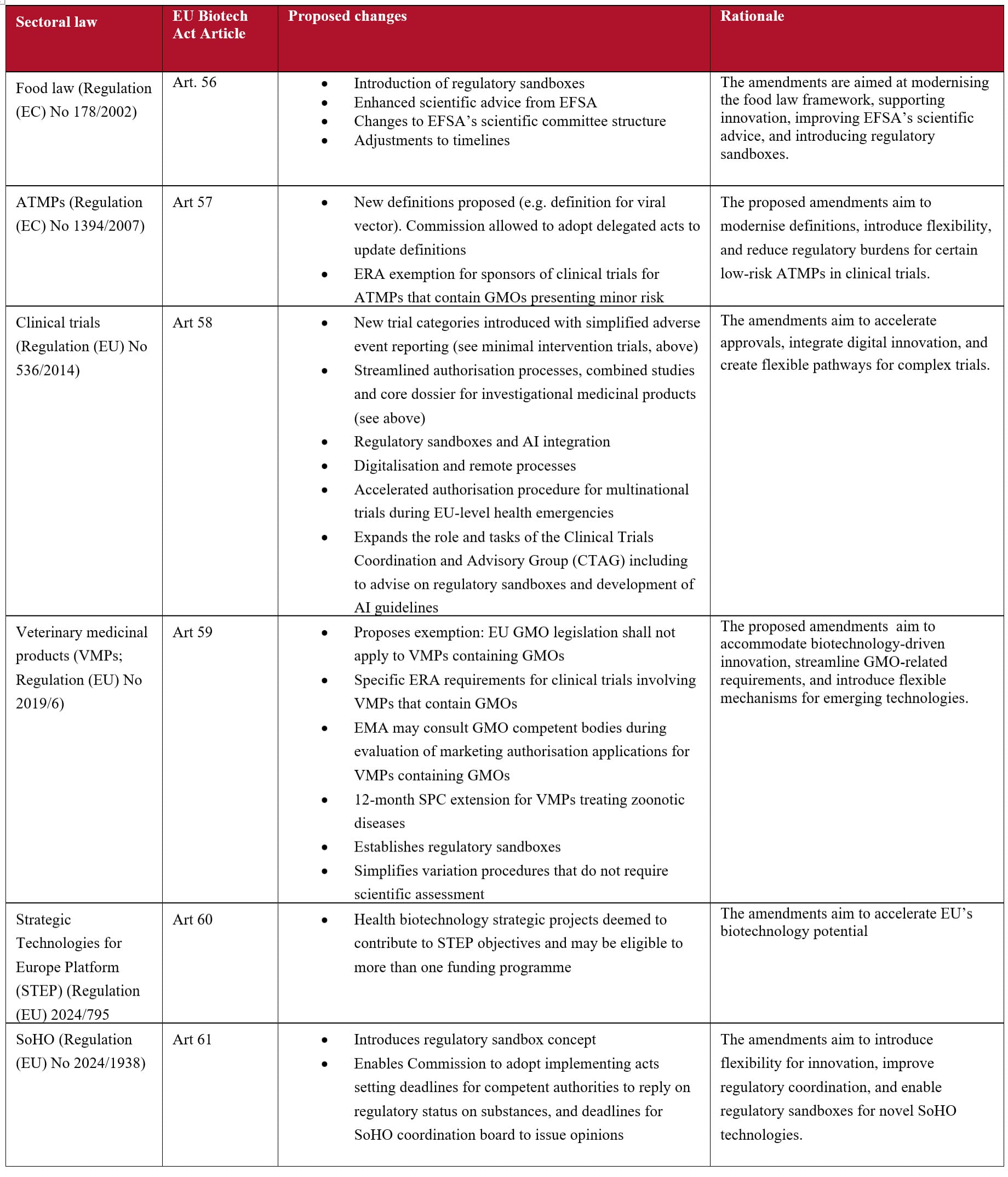
The Act covers the entire lifecycle of health biotechnology products and services, from initial research and funding, to development, manufacturing and market access. The text spans 67 Articles and 3 Annexes and it includes proposals to amend a number of EU regulations, in particular:
- Regulation (EU) No 536/2014 on clinical trials
- Regulation (EC) No 1394/2007 on advanced therapy medicinal products (ATMPs)
- Regulation (EU) 2024/1938 on standards of quality and safety for substances of human origin (SoHO Regulation)
- Regulation (EU) 2019/6 on veterinary medicinal products
- Regulation (EC) No 178/2002 on general principles and requirements of food law
- Directive 2001/18/EC on genetically modified organisms (GMOs)
- Regulation (EU) 2024/795 establishing the Strategic Technologies for Europe Platform (STEP Regulation)
- Regulation (EC) No 469/2009 on supplementary protection certificate (SPCs)
Proposals of this scale are normally preceded by a full impact assessment to ensure transparency and evidence-based policymaking. However, citing “political urgency”, the Commission has bypassed this step in this case opting instead for an analytical staff working document. Some stakeholders have expressed their concern that this approach may have been rushed, leaving limited time to explore alternatives and assess long-term implications of the future Regulation. A second legislative proposal, expected in Q3 2026, will cover the wider biotech ecosystem including agriculture, energy, defence, and industrial biotech sectors.
Below is an overview of some of the key measures foreseen in the Commission’s Proposal for a European Biotech Act:
Accelerating the EU’s biotechnology potential through targeted funding
The Act establishes a framework for the recognition and support of ‘health biotechnology strategic projects’ and ‘high-impact strategic projects’ conducted in the EU. These projects will benefit from accelerated procedures, administrative and technical support, and priority access to funding. The Act also promotes cooperation across networks of health biotechnology clusters to upscale research and facilitate the transfer of knowledge. The overall aim of these provisions is to ensure that innovation translates into creation of European jobs and an expansion of Europe’s bio-manufacturing capacities.
A centrepiece of the Act is the creation of an EU Health Biotechnology Investment Pilot, developed in partnership with the European Investment Bank Group. Designed to bridge the current funding gap for biotech companies, especially at the scale-up and late-stage development phases, the Pilot brings together public and private capital, offering tailored equity and venture debt instruments, and supporting companies throughout their lifecycle.
12-month SPC extension to protect innovation
Importantly, the Act proposes a 12‑month extension to the existing Supplementary Protection Certificate (SPC) for biotech medicinal products and advanced therapy medicinal products (ATMPs) that meet all of the following conditions set out in Article 27(1):
- the medicinal product contains a new active substance “distinctly different” from that of any authorised medicinal product in the Union;
- the medicinal product has a mechanism of action “distinctly different” and shows a level of safety and efficacy which is at least equivalent to that of any authorised medicinal product in the Union for the same disease;
- the clinical trials evaluating the efficacy of the medicinal product and supporting its marketing authorisation were conducted in more than two Member States;
- at least a manufacturing step, excluding packaging, quality testing and certification is performed in the Union.
This measure is designed to incentivise onshore development and manufacturing of cutting-edge biologics in the EU. While industry groups view it as a boost for competitiveness, health authorities have raised concerns about its potential impact on healthcare budgets. This will certainly be a debated and sensitive measure during Council and EP deliberations. Clearer criteria should help identify which biotech medicinal products and ATMPs are concerned, especially since the Act does not define what qualifies as “distinctly different”.
The Act also proposes a 12-month SPC extension for veterinary medicinal products aimed at treating zoonotic diseases (see table below).
Speeding up Clinical Trials and Market Access
To strengthen the EU’s attractiveness for clinical research, the Act proposes amendments to the Clinical Trials Regulation aimed at streamlining and modernising clinical trial processes. Key changes include:
- New category of ‘minimal-intervention clinical trials’: This applies to investigational medicinal products that are already authorised, used in accordance with the terms of the marketing authorisation, and pose minimum risk to patient safety. Both minimal and low intervention trials will benefit from simplified procedures.
- Single core dossier for investigational medicinal products: The Act allows sponsors to create and reference a single core dossier across all associated clinical trials, reducing duplication and accelerating development and regulatory review.
- A single authorisation process for combined studies involving a medicine and a medical device or an in-vitro diagnostic which is under a clinical investigation or performance study.
- Regulatory sandboxes: Controlled environments will be introduced to support innovative trial designs and the use of digital or AI-enabled approaches.
- Shortened timelines: The Act proposes to reduce the authorisation period for multinational clinical trials from 106 days to 75 days, with a further cut to 47 days where no additional information is required from the sponsor.
Additional proposed changes are outlined in the table below, which provides a summary of amendments to the Clinical Trials Regulation and other related sectoral regulations.
AI and data as biotechnology enablers
The Act introduces a series of measures to integrate advanced technologies, including AI, into the biotechnology ecosystem and strengthen data infrastructure for innovation. These measures seek to enable safe and effective deployment of AI in biotech, improve data quality for advanced analytics, and create controlled environments for innovation. Alignment with the European Health Data Space, the Digital Omnibus package and other Union data initiatives will be crucial.
Regulatory tools for novel biotech products
The Act also dedicates an entire chapter to mechanisms designed to help developers navigate the complex regulatory frameworks available for innovative health biotechnology products and services, and to enable controlled testing environments (i.e. regulatory sandboxes) for novel solutions. Key measures include assisting developers in identifying the appropriate regulatory pathways for products that raise questions about the applicability of existing frameworks (e.g. medical devices, SoHO, ATMP frameworks…) and providing guidance on combined products and access to regulatory sandboxes. This advice will be provided by the EU Health Biotechnology Support Network. This Network will be set up and coordinated by the European Commission with support from contact points or units (“antennas”) located in every member state.
Biodefence and preventing biotechnology misuse
This Act foresees measures to strengthen EU biodefence capabilities, enhance biosecurity, and prevent misuse of biotechnology, including risks linked to advanced technologies such as AI. The objective is to build a resilient EU biodefence system, ensure responsible innovation, and prevent misuse of biotechnology through strict controls, transparency, and cross-sector collaboration
Simplifying the EU’s regulatory landscape
Finally, and as noted above, an important aspect of the EU Biotech Act is the fact that it proposes to amend multiple existing regulations. These changes are intended to streamline regulatory processes, reduce administrative complexity, and create unified pathways for complex and innovative products. The table below lists the most significant amendments:
A Vision for a World-Leading European Biotech Industry?
The EU Biotech Act represents a comprehensive legislative proposal with the potential to significantly influence Europe’s health biotechnology sector. Its primary purpose is to create an environment that encourage companies to conduct research, development, and manufacturing within the EU. Given its breadth and complexity, the proposal will require detailed analysis and is expected to spark significant debate in the European Parliament and Council especially on sensitive issues such as intellectual property incentives and funding priorities.
The text is complex and will require careful legal and policy analysis, particularly given its interaction with other major legislative initiatives such as the EU Pharma Package and the evolving regulatory frameworks for AI and data governance. These interdependencies will be critical to assess, as they shape compliance obligations, timelines, and strategic planning for industry stakeholders. How these discussions unfold will be key to watch as we move towards 2026.




